Chapter 2: Matter and Change
|
Section 2.3: Elements and Compounds
Our textbook waits until Chapter 5 to introduce the periodic table, but this well-known table is an organized list of the elements. That makes it a great tool for thinking about the difference between elements and compounds!
Use the links below to print a periodic table. These printable and clickable periodic tables are from the Thomas Jefferson National Accelerator Facility in Virginia. In addition to doing basic research on atoms, they have interesting resources about chemistry, physics, and their facility. Check them out here: https://www.jlab.org/. |
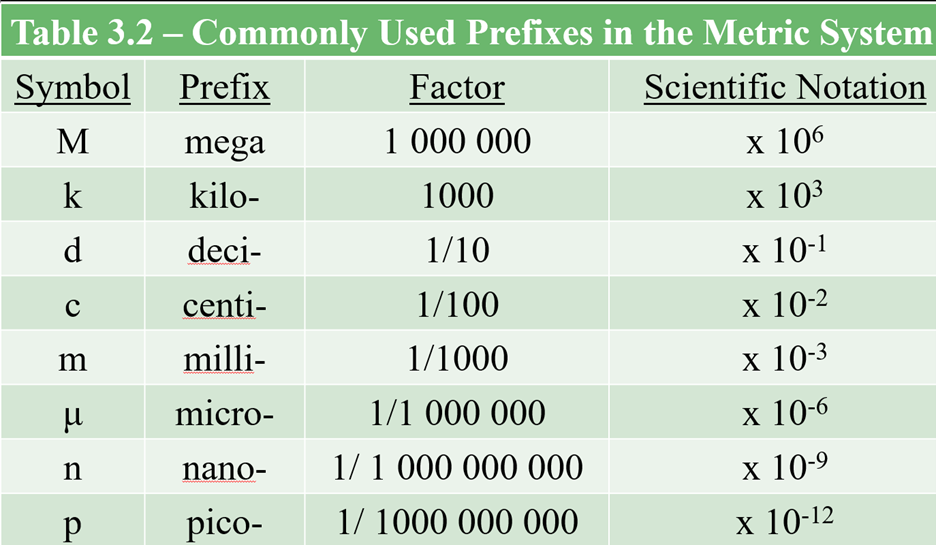
Chapter 3: Scientific Measurement
|
|
Significant Figures Step by Step
Melissa Maribel explains the rules for how to identify the significant digits of numbers and how to consider significant figures when adding, subtracting, multiplying, or dividing. If you want a slower, more deliberate explanation of significant figures, Tyler DeWitt has you covered! |
|
|
Why are Significant Figures Important?
Tyler DeWitt explains the "why" behind significant figures. Scientific Notation: Multiplication and Division
Tyler DeWitt explains how to multiply and divide numbers written in scientific notation. If you are looking for the basics of scientific notation, here is his introduction to scientific notation. Metric Prefixes
For quizzes and exams throughout the year, you are expected to memorize the following prefixes: mega-, kilo-, centi-, milli-, and micro-. |
Chapter 4: Problem Solving in Chemistry
|
|
Converting Units with Conversion Factors
Tyler DeWitt has videos that start out basic and work up to more complex unit conversions. In this video, he explains conversions with a single conversion factor. Converting Units Using Multiple Conversion Factors
In this video, Tyler DeWitt explains unit conversions with multiple factors. |
Chapter 5: Atomic Structure and the Periodic Table
|
|
What's the Difference Between Mass Number and Atomic Mass?
Tyler DeWitt gives a great explanation. |
Chapter 6: Chemical Names and Formulas
Error in homework: 1.d. should say "calcium atom" instead of "calcium ion."
For more Chapter 6 resources, see Quarter 2.