|
Below are two printable periodic tables and a clickable periodic table all from the Thomas Jefferson National Accelerator Facility in Virginia. In addition to doing basic research on atoms, they have interesting resources about chemistry, physics, and their facility. Check them out here: https://www.jlab.org/.
Chapter 6: Chemical Names and Formulas
Error in homework: 1.d. should say "calcium atom" instead of "calcium ion."
In the short video below, Dr. Breslyn explains how to name ionic and molecular compounds--and how to tell if a compound is ionic or molecular. For a list of all his videos about naming and writing formulas, click here. (You can skip the videos about naming acids, since I am not requiring you to be able to name acids.)
|
|
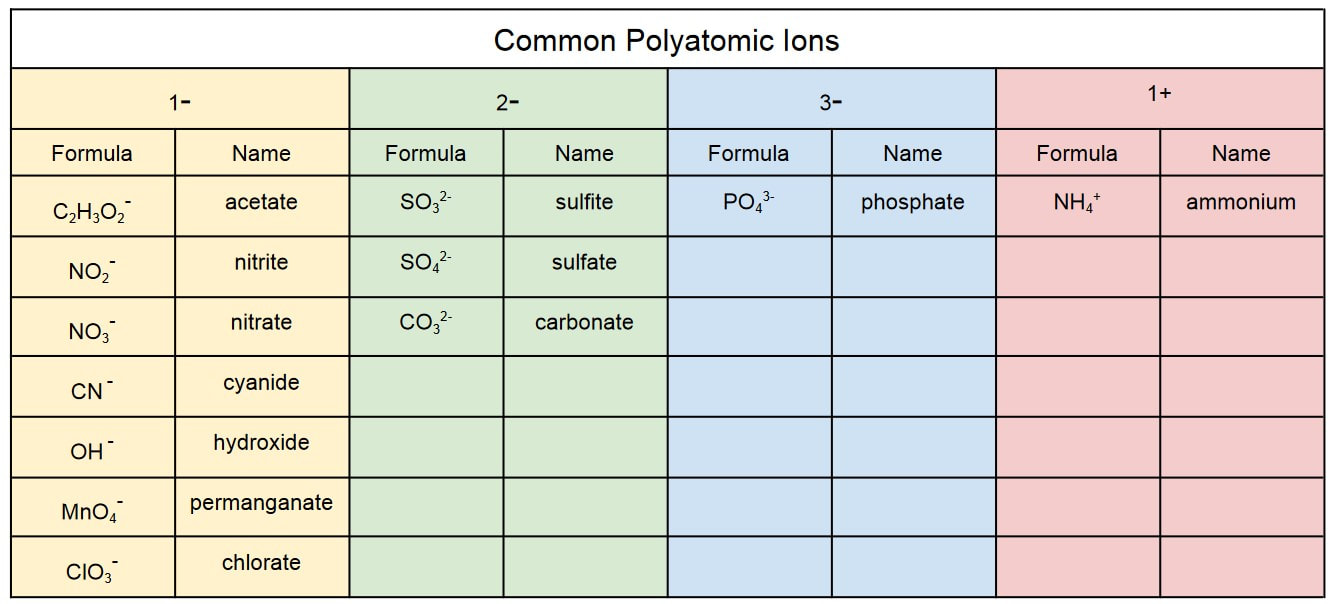
Tyler DeWitt explains how to write formulas with polyatomic ions. When a formula has more than one of the same polyatomic ion, you will need to use parentheses. Watch this video for examples.
The chart below lists the polyatomic ions you will be responsible for knowing on quizzes and tests throughout the rest of this class.
|
|
Looking for another way to learn about naming ionic compounds and writing their formulas? Khan Academy has a helpful series of articles, videos, and practice problems. Click on this link to get started with "Names and formulas of ionic compounds." In the section "Polyatomic ions," you should probably skip a couple of paragraphs titled "Structure of polyatomic ions," which go into more detail than you need. The rest will reinforce what you have read in Chapter 6.
|
Chapter 7: Chemical Quantities
|
Need help with Chapter 7? Tyler DeWitt has created a great series of videos to explain moles! The first video is below, and here is a link that will take you to his entire playlist of eleven videos on moles. If you already know the basics, you may want to increase the playback speed or even skip ahead to topics you find more challenging.
|
|
Dr. Andrew Robertson explains the history of Avogadro's Number, starting with Democritus in ancient Greece and moving forward through the experiments of many chemists and physicists during the last few centuries. Watch this if you want to gain a better understanding of how science develops with time. Section 7.3: Percent Composition and Chemical Formulas
|
|
Tyler DeWitt explains (1) how to calculate the percent composition by mass and (2) how to avoid common mistakes in these calculations.
|
|
|
Empirical Formula
Socratica's Empirical Formula video shows how to find the empirical formula based on the percent composition. |
Chapter 8: Chemical Reactions
|
Section 8.1: Describing Chemical Change
|
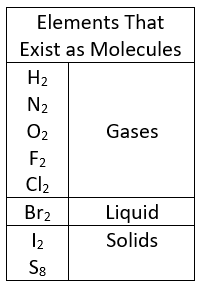
For the curious, here is a little more information about sulfur, which exists in many forms but most commonly as a ring of eight sulfur atoms per molecule.
|
These four videos explain balancing chemical equations. The Tyler Dewitt videos (the top two) start with the basics and move slowly. The Wayne Breslyn videos (the bottom two) move more quickly and explain balancing chemical equations involving polyatomic ions.
|
|
|
|
Section 8.2: Types of Chemical Reactions
If you want a video review of types of reactions, this video has the basics of the reactions in Section 8.2.
|
|
The two videos below explain how to predict if a single replacement reaction will occur. The first one is about more reactive metals replacing other metals. The second video is about more reactive halogens replacing other halogens. Near the end of the second video, Dr. Breslyn explains another type of reaction, redox reactions, that you are not responsible for knowing about in this class.
|
Section 8.3: Reactions in Aqueous Solution
We will watch the first of these videos in class. The second video will guide you through several examples on how to write net ionic equations. Dr. Breslyn always balances the chemical equation in the first step, while I encourage you to make it easier on yourself and wait until the last step to balance the net ionic equation. He includes some examples with weak acids and bases that do not separate into ions in solution. You do NOT need to be able to recognize weak acids and bases and can assume that all aqueous compounds separate into their ions.
|
Table 8.3 in our textbook lists the solubility rules for ionic compounds. You will use these rules to predict when a precipitate will form. The table has two errors. Rules 1 and 5 mention ammonia when they should say ammonium ion. Ammonia is NH3 (a molecular compound). Ammonium ion is NH4+ (a polyatomic ion you should have memorized).
The video below explains solubility rules. As Wayne Breslyn says, some instructors would require students to memorize the solubility rules. You are NOT expected to memorize the solubility rules, but you should be able to use the rules to predict solubility. A compound will precipitate (fall to the bottom of its container as a solid) if it is insoluble. Note that precipitate is both a noun and a verb. Precipitates are compounds that precipitate.
Oxalates are not included in our book's solubility rules, but there are at least two reasons that their insolubility has practical importance. Calcium oxalate is what makes up most kidney stones. Calcium oxalate is also the mineral that makes certain plants (e.g., calla lily and philodendron) toxic. This can be an issue for pets and small children.
|
|
Chapter 9: Stoichiometry
Stoichiometry is the process of calculating the quantities of reactants and products in chemical reactions. The word stoichiometry comes from the Greek word stoikheion, which means "element," and the suffix -metry, which means "process of measuring," so stoichiometry literally translates to "the process of measuring elements."
Section 9.2: Chemical Calculations
|
The Mole: Avogadro's Number and Stoichiometry
This video from Professor Dave Explains starts by explaining moles and at 2:43 begins to explain stoichiometry. He even has practice problems built into the video! |