Chapter 13: Electrons in Atoms
|
|
Writing Electron Configurations
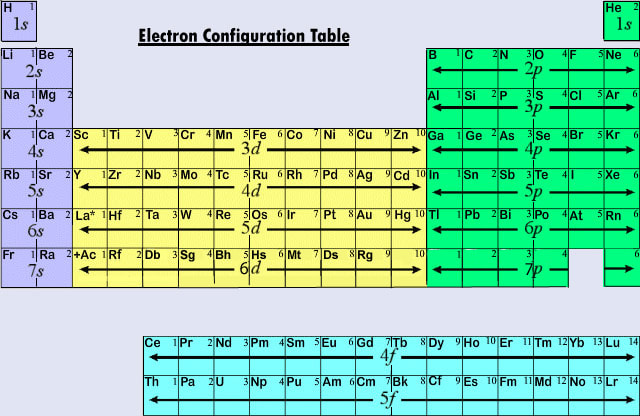
The Electron Configuration Table to the left is from an article in LibreTexts Chemistry. It gives a great visual for how electron configurations relate to the periodic table. The Dr. B video below it walks you through the same information. Here is a link to Dummies.com, which includes the chart I showed in class for remembering the order that sub-levels are filled with electrons. (Other sources may show a flipped version of this chart, but I like how this version shows the lowest energy sublevels at the lowest position.) You should memorize this chart and/or know how to use the periodic table so that you can write electron configurations. |
|
|
A Better Way to Picture Atoms
If you want another explanation for what atomic orbitals looks like, check out these cool animations! |
|
|
Quantum Mechanics and the Schrödinger Equation
What does it mean for an electron to be a wave? Professor Dave explains . This is an optional video that goes beyond what you need to know for this class. |
Chapter 14: Chemical Periodicity
Chapter 15: Ionic Bonding and Ionic Compounds
|
|
Ionic Bonding
These three Tyler DeWitt videos are integrated with your homework for Chapter 15. |
|
|
What Are Metallic Bonds?
This video has a simple explanation of metallic bonds and how the properties of metals relate to their bonds. |
|
|
Bond Types
High school chemistry teacher Nick Duell gives an overview of types of bonds. His explanation of metallic bonds is very brief. |
|
|
Metallic Bonds
Professor Dave explains metallic bonds. He goes a little deeper than our textbook. |
Chapter 16: Covalent Bonding
|
For the Shapes of Molecules worksheet, do not sketch the shape of the molecule. Instead, draw the Lewis Dot Structure and then name the shape of the molecule. This worksheet is to be completed in class. |
Section 16.1: The Nature of Covalent Bonding
Section 16.2: Bonding Theories
Section 16.3: Polar Bonds and Molecules
Chapter 18: Solutions
|
Section 18.1: Properties of Solutions
Section 18.2: Concentrations of Solutions
Section 18.3: Colligative Properties of Solutions
Section 18.4: Calculations Involving Colligative Properties
|
Section 18.4 with Mrs. Morales
My predecessor at PEP, Mrs. Morales, recorded lessons for this chapter. You may find these helpful. For 18.4, we are not covering mole fractions, which she explains in 8:15 to 13:15. Later in the video, she uses a slightly different method than our textbook for calculating boiling point elevation and freezing point depression. Rather than calculating effective molality, she adds the Van't Hoff factor to her equations. I'll be using the book's method. |